Our services
Comprehensive GMP engineering and consulting solutions for pharmaceutical and biotech facilities
GMP engineering services provide technical oversight, regulatory compliance, and design expertise for pharmaceutical facilities. By integrating owner representative services with detailed engineering from concept to commissioning, these services ensure facilities meet PIC/S and FDA standards , significantly reducing operational risks. Get more technical introductions 「The Strategic Blueprint for High-Performance Pharma & Biotech Facilities」
The Core Pillars of GMP Engineering
Identifying potential compliance gaps during the design phase prevents costly retrofits later in the project lifecycle.
Detailed engineering and vendor management ensure budgets are maintained through rigorous change control processes.
Integrating commissioning and validation (C&V) early prevents delays during the critical final startup phase.
The Owner Representative Role
An owner representative acts as your technical advocate, ensuring that the design team, contractors, and vendors fulfill their contractual obligations. In the context of GMP facility design, they ensure technical specifications align perfectly with User Requirement Specifications (URS).
Specialized Cell Therapy Requirements
Maximizing closed processing to eliminate environmental contamination risks in patient-specific manufacturing.
Adhering to 21 CFR Part 11 and ALCOA+ principles for robust Computer System Validation and data integrity.
Key Takeaways for Success
To optimize ROI, engage GMP specialists during the conceptual design phase. Prioritize validation as a core construction activity rather than an afterthought, and utilize an owner representative to maintain strict quality control over technical vendors.
Owner Representative Services

We act as your representative on-site, protecting your interests throughout the project lifecycle. Our team monitors design quality, construction progress, equipment procurement, and ensures regulatory compliance.
GMP Facility Design
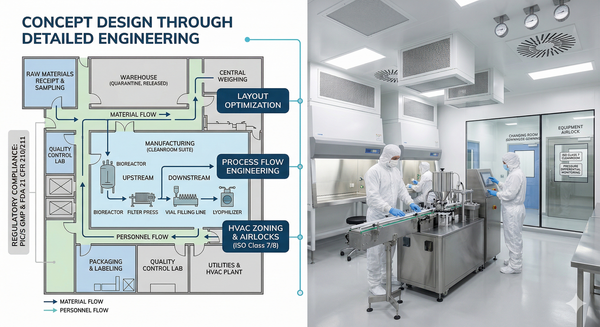
From concept design through detailed engineering, we ensure your facility meets PIC/S GMP and FDA requirements. Our expertise covers layout optimization, HVAC systems, cleanroom design, and process flow engineering.
Computer System Validation (CSV)
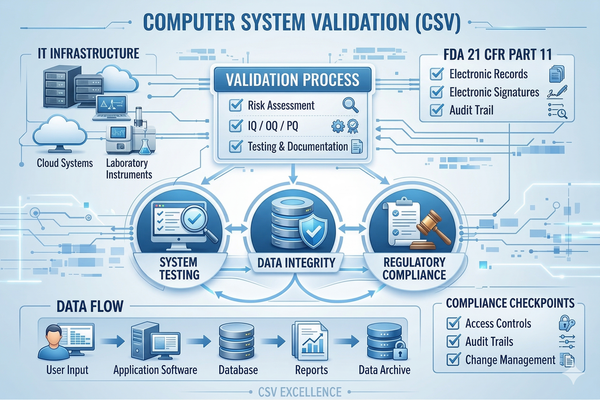
Ensure your critical manufacturing systems comply with FDA 21 CFR Part 11. We provide comprehensive CSV consulting including risk assessment, IQ/OQ/PQ protocols, and documentation.
Project Management & Commissioning
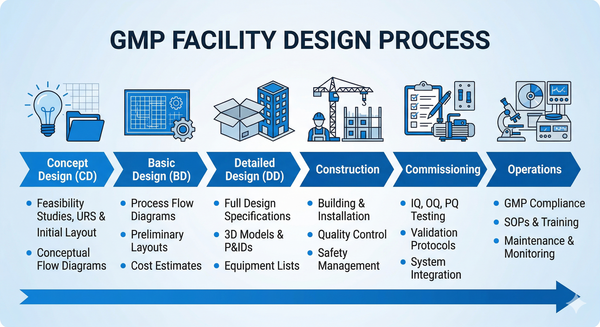
Comprehensive project management from planning through commissioning. We optimize schedules, control budgets, manage vendors, and ensure smooth facility startup and operations.
Strategic Engineering AI Advisor
Delivering cross-disciplinary technical insights and comprehensive consultancy for regulated industries. Access real-time guidance on engineering excellence, compliance frameworks, and lifecycle management.
Initialize consultation via the lower right portal


